Donation after circulatory death (DCD) represents a large pool of donors for lung transplantation (LTx). Currently, suitable DCD donors used by our institution include patients who are having withdrawal of life-sustaining treatment (WLST) based on futility, with death expected within 90 minutes of withdrawal of active treatment, and donation after brain death (DBD) patients who go into cardiac arrest before planned donation (Maastricht category III and IV patients). Standard contraindications to lung donation apply. Prior cardiac surgery is not an absolute contraindication in our experience. Specifically, patients who are not in an intensive care unit (ICU) or those in whom death is highly likely to occur more than 90 minutes after WLST are not considered suitable for DCD donation. Consent for donation is sought from next of kin by a ICU medical specialist. Following consent, specific tests such as blood for serology, cross matching, and arterial gases are obtained to assess the suitability of the donor lungs and allow matching with potential recipients. Invasive tests such as heparin administration and bronchoscopy may or may not be permitted before WLST or death depending on local institutional guidelines.
The operating room (OR) staff, along with the thoracic and abdominal organ retrieval teams, are notified 4 hours before the withdrawal of active treatment in a potential donor. Approximately 30 minutes before WLST, a meeting of the organ retrieval teams is held in the OR complex to discuss the clinical details of the potential donor and the logistics involved in organ procurement. ICU staff manage the patient and WLST (typically by removal of the endotracheal tube) as per local standards of practice. Death is declared by an ICU specialist on cessation of circulation for 5 minutes, as evidenced by the absence of pulse on arterial trace for 5 minutes with or without the absence of electrical activity on electrocardiogram monitoring. The donor is transported expeditiously to the OR, ideally within 10 minutes of the declaration of death.
All steps from the transport of the patient from the ICU to cannulation of the pulmonary artery are standardized, as shown in the section Technique, to institute pulmonary flush solution as soon as practical. However, there is evidence that there is a safe margin of at least 60 minutes following a donor systolic blood pressure being less than 50 mm Hg for administration of pulmonary flush. The surgical technique is similar in principle to that of the well-established lung donation technique from brain-dead donors .
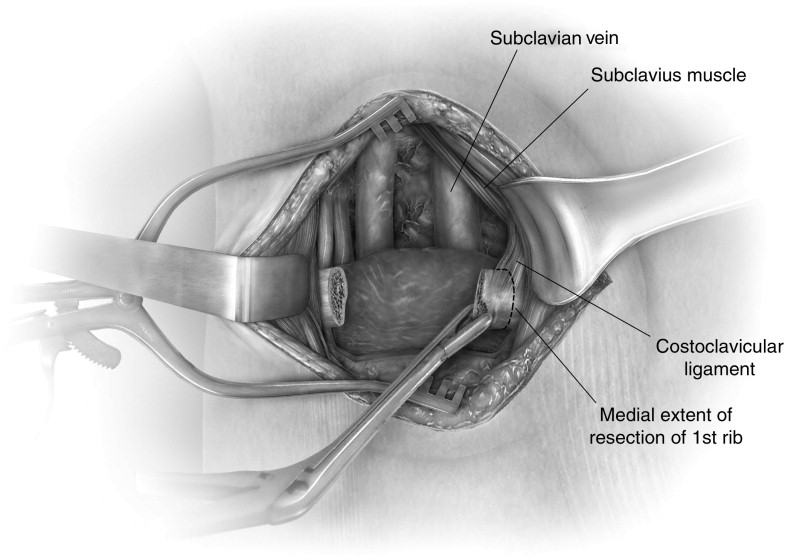
Technique
(Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10, Figure 11, Figure 12)












Discussion
Between May 2006 and July 2014, there have been 240 DCD donor referrals to our institution, resulting in 124 surgical team attendances attempting to retrieve donor organs. Overall, 91 donors proceeded on to cardiac arrest and provided suitable lungs for 92 DCD LTx. These DCD numbers represent 19% of 487 LTx procedure performed at the Alfred hospital over this time. DCD LTx had added 23% more transplants and contributed significantly to decreasing the waiting list mortality from 29% to 5% over this period (P < 0.05).
Early outcomes following DCD LTx were similar to the contemporaneous DBD donors. There was 1 death from sepsis day 16 (99% survival at 30 days) and a further 3 deaths in the first year from fibrinoid organizing pneumonia day 56, renal failure day 124, and poor compliance day 351 (94% survival at 1 year). Primary graft dysfunction, airway ischemia, and acute rejection were the same in both DCD and DBD LTx. Long-term outcomes were excellent with 5- and 8-year survival rates of 71% in the DCD group compared with 58% and 46%, respectively, in the DBD group (P = 0.08). Our protocol and techniques of lung procurement have helped develop a national DCD LTx program in Australia.1
Conclusion
The use of DCD category III and IV donor lungs for LTx makes significant inroads into the worldwide shortage of donor lungs and waiting list mortality. A detailed protocol-based approach to the recognition, assessment, and techniques of retrieval of DCD lungs is critical. In turn, this contributes to minimizing wasted retrieval attempts where the donor does not undergo arrest and maximizes clinical outcomes. The short- and long-term outcomes are excellent (notably without ex vivo lung perfusion) and justify a recommendation that all LTx centers should, at least, explore their DCD donor pool using the techniques we have described.
原文链接:http://www.xxwk.net/archives/1986