Baranov NS, Slootmans C, van Workum F, Klarenbeek BR, Schoon Y, Rosman C. Outcomes of curative esophageal cancer surgery in elderly: A meta-analysis. World J Gastrointest Oncol 2021; 13(2): 131-146
背景
越来越多的老年患者正在接受食道癌的根治性手术治疗。先前的荟萃分析表明,与年轻患者相比,老年患者的术后发病率和死亡率更高,这可能导致根据年龄选择患者。然而,仅包括接受开放食管切除术的患者的研究。因此,尚不清楚接受微创食管切除术的患者的年龄与预后是否相关。
目的
对接受食管切除术的食管癌患者的年龄和术后结果进行系统评价。
方法
纳入比较老年和年轻原发性食管癌患者接受根治性食管切除术的研究。手稿中介绍了使用 75 岁年龄阈值的研究的荟萃分析,补充材料中使用其他年龄阈值的研究。在 MEDLINE、Embase 和 Cochrane 图书馆中搜索了 1995 年至 2020 年间发表的文章。使用纽卡斯尔-渥太华量表评估偏倚风险。主要结局是吻合口漏、肺和心脏并发症、谵妄、30 天和 90 天以及住院死亡率。次要结局包括肺炎和 5 年总生存率。
结果
七项研究(4847 名患者)使用 75 岁的年龄阈值进行荟萃分析,其中 755 名老年患者和 4092 名年轻患者。与年轻患者(3.92%)相比,老年患者(9.05%)的 90 天死亡率更高(置信区间 = 1.10-5.56)。此外,与年轻患者(3.68%)相比,老年患者(9.45%)的住院死亡率更高(置信区间=1.01-5.91)。在 2 项微创食管切除术研究的亚组中,老年和年轻患者的 30 天、90 天和住院死亡率相当。
结论
接受食管癌根治性食管切除术的老年患者术后死亡风险较高。微创食管切除术对于降低老年患者的死亡率可能很重要。
INTRODUCTION
Since the incidence of esophageal cancer is rising as well as the average age of the global population, more older patients are expected to be diagnosed with esophageal cancer[1,2]. In the West, already around 30 percent of patients undergoing curative esophagectomy for esophageal cancer is aged 70 years or older[3]. Increased use worldwide of minimally invasive esophagectomy (MIE) may benefit these older patients because it is associated with lower postoperative morbidity[4-7]. However, patients aged 75 or 80 years or older were excluded from all major trials that compared MIE with open esophagectomy (OE)[4-6].
One meta-analysis by Markar et al[8] from 2012 comparing OE outcome between older and younger patients showed that older patients had a higher rate of in-hospital mortality, pulmonary and cardiac complications and a lower 5-year overall survival. Two recent meta-analyses from 2019 and 2020, which included only studies using an age threshold of 80 years to distinguish old from young, reported similar results[9,10]. However, these three meta-analyses did not include studies on MIE, and excluded studies that used other age thresholds. In contrast, recent individual studies that compared older with younger patients who underwent MIE showed comparable rates of anastomotic leak as well as 30-d mortality[11-13]. Regarding pulmonary and cardiac complications, however, results are conflicting. No systematic review of recent literature including MIE studies that compared older with younger patients, has been undertaken to this date. Therefore, it remains unknown whether MIE could be beneficial to older patients regarding postoperative morbidity and mortality.
We aimed to investigate the association between patient age and outcome after curative (open and minimally invasive) esophagectomy for esophageal cancer.Go to:
MATERIALS AND METHODS
The protocol for this review was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under number CRD 42019121754. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline was followed[14].
Eligibility criteria
The inclusion criteria were studies comparing older with younger patients undergoing esophagectomy with curative intent for primary esophageal cancer (regardless of age threshold). The exclusion criteria were: Studies with more than 20% stage IV patients, salvage or palliative esophagectomy, conference abstracts, cross-sectional studies, case series, case reports and letters to editors. Studies having more than two age groups were included only when it was possible to combine the age groups in an older and younger age group.
Information sources
The electronic databases of MEDLINE, Embase (both through the Ovid interface) and The Cochrane Central Register of Controlled Trials were systematically searched. Reference lists from the included studies were also searched. The search strategy was composed in collaboration with a librarian from the Radboud University Medical Library.
Search
The MEDLINE search strategy was: [exp ESOPHAGECTOMY/ or (Esophagectom* or Oesophagectom* or Esophagus or Oesophagus or Oesophageal or Esophageal) and (Resection* or Surger* or Laparoscop* or Thoracoscop*).ti,ab,kf.] AND (exp Esophageal Neoplasms/ or (Esophag* or oesophag*) and (cancer* or neoplasm* or carcinoma* or adenocarcinoma* or malignanc*).ti,ab,kf.) AND (Aged or Old* or Frail*).ti,kf. or (Older* or Elder* or Senior* or Geriatric).ti,ab,kf or Age Factors/ or exp *AGED/ or exp AGE DISTRIBUTION/. Comparable search strategies were used in Embase and the Cochrane Library. Complete search strategies were listed in the protocol for this review. No language restrictions were applied and all results up to January 1st, 2020 were included.
Study selection
First, two reviewers (NB and CS) independently screened titles and abstracts for potentially relevant studies. Second, two reviewers (NB and CS) independently examined the full text of potentially relevant studies for eligibility. When disagreement occurred during this phase, a third reviewer (FvW) was consulted until consensus was reached. Two reviewers (NB and CS) screened reference lists from the included studies for potentially relevant articles repeating aforementioned processes when articles were deemed eligible.
Data collection
When possible, data of the studies included in the systematic review were pooled for quantitative meta-analysis. Otherwise, the data was described. For the purpose of this review the following data was extracted: Patient and tumor characteristics, surgical technique and approach, operation characteristics, complications, hospital stay, mortality, survival and quality of life.
Risk of bias in individual studies
The Newcastle-Ottawa quality assessment scale was used to assess bias in studies included in this review[15]. This scale rates studies on three sources of bias based on eight criteria. Each criterion is worth one star except confounding, which is worth two stars. For this systematic review, studies scoring 7-9 stars were considered to be of high methodological quality, studies scoring 4-6 stars were considered to be of moderate methodological quality, and studies scoring 1-3 stars were considered to be of low methodological quality.
Outcome measures
The primary outcomes were: The rate of anastomotic leak, pulmonary and cardiac complications, delirium, 30-d, 90-d and in-hospital mortality. After multiple thorough discussions between the authors, medical doctors from both the department of surgery and geriatrics, these outcomes were deemed most clinically relevant to answer our research question.
The secondary outcomes were: the rate of chylothorax, pneumonia, hospital length of stay, quality of life, 5-year survival and 5-year disease-specific survival.
Risk of publication bias across studies
To assess publication bias, funnel plots with the effect measures on the x-axis and standard error on the y-axis were used for visualization and the Egger’s test was used for quantification. Funnel plots and Egger’s tests were performed for the primary outcome measures.
Age threshold
It was decided, after careful deliberation among the authors, to present in the current article the meta-analysis results of studies using an age threshold of 75 years, because it was deemed most relevant for current surgical practice, and prominent randomized controlled trials (TIME, MIRO) used this age threshold as an exclusion criterion[4,5]. Meta-analysis results of studies using other age thresholds as well as all age thresholds combined were presented in the supplementary only (Supplementary Table 1).
Synthesis of results
A quantitative synthesis of aggregate patient data (for OE, MIE and both groups together) was performed. Regardless of study homogeneity in terms of design and comparators and regardless of statistical homogeneity as expressed by the I², we conducted a meta-analysis using a random-effects model. Dichotomous data were analyzed by using relative risks with a 95% confidence interval (CI). Continuous outcomes were analyzed using weighted mean differences (with a 95%CI) or standardized mean differences (95%CI) if different measurement scales were used. If data was reported as median with ranges (maximum, minimum), the formula described by Wan et al[16] was used to estimate the mean, variance and standard deviation. If inter-quartile ranges were reported without minima and maxima, the data were presented descriptively when deemed necessary. If desired data was missing, authors were contacted when deemed necessary. The data was synthesized using the appropriate imputation methods, otherwise the data was presented descriptively. Statistical heterogeneity was tested using the Chi² test (significance level: 0.1) and I² statistic (0% to 40%: Might not be important; 30% to 60%: May represent moderate heterogeneity; 50% to 90%: May represent substantial heterogeneity; 75% to 100%: Considerable heterogeneity). Outcomes were combined and calculated using the statistical software program R with the package “meta” in accordance with Doing Meta-Analysis in R by Harrer et al[17-19].Go to:
RESULTS
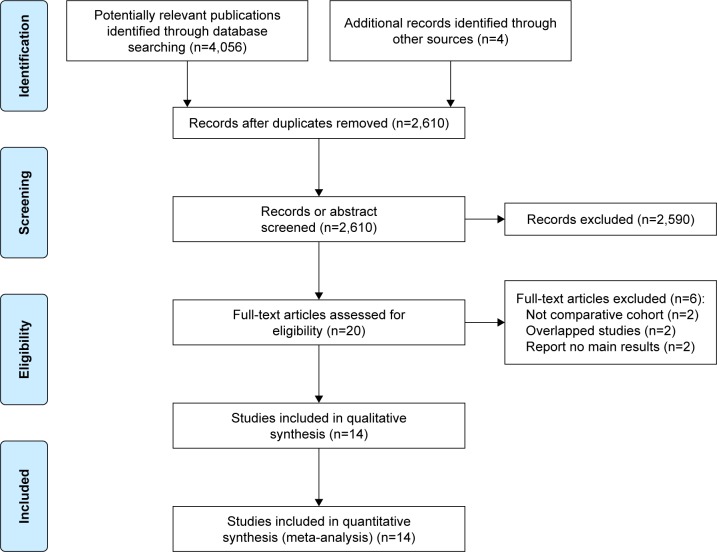
Figure Figure11 shows the study selection process. A total of 3647 records were identified through database searching and cross-referencing. Overall, 8 studies were included for qualitative synthesis of which 7 studies were included for quantitative analysis (meta-analysis) with 4991 and 4847 patients respectively[13,20-26].

Open in a separate windowFigure 1
Flow chart of study selection. aReasons for exclusion: other age threshold: 49; benign disease: 1; case series: 1; conference abstract: 38; duplicate: 4; letter to the editor: 2; no comparison between older and younger patients: 28; non-surgical treatments: 3; other cancer types: 2; overlapping study cohorts: 3; review: 2; too many palliative patients: 20; unclear study design: 4.
Study characteristics
One study was published in 2009 and the rest was published after 2010. Regarding operative approach, 2 studies were deemed as “MIE” (> 50% MIE), 3 as “OE”, and 3 as “Unknown” (studies of whom the surgical approach could not be determined).
Risk of bias
From the 7 included studies for meta-analysis, 2 were of “High Methodological Quality”, 5 were of “Medium Methodological Quality” and none were of “Low Methodological Quality”. Individual studies’ star counts are shown in Table Table11.
Table 1
Risk of bias per included study for meta-analysis
| Ref. | Age | A | B | C | D | E | F | G | H | Stars |
| Akutsu et al[20] | 75 | 1 | - | 1 | - | 1 | 1 | 1 | - | 5 |
| Baranov et al[13] | 75 | 1 | 1 | 1 | - | 1 | 1 | 1 | - | 6 |
| Klevebro et al[22] | 75 | 1 | 1 | 1 | 1 | - | 1 | 1 | - | 6 |
| Li et al[23] | 75 | 1 | 1 | 1 | 1 | - | 1 | 1 | - | 6 |
| Schweigert et al[25] | 75 | 1 | 1 | 1 | 1 | - | 1 | 1 | - | 6 |
| Yang et al[26] | 75 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | - | 7 |
| Kanda et al[21] | 75 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | - | 7 |
A: Representativeness of the exposed cohort; B: Selection of the non-exposed cohort; C: Ascertainment of exposure; D: Demonstration that outcome of interest was not present at start of study; E: Comparability of cohorts on the basis of the design or analysis; F: Assessment of outcome; G: Was follow-up long enough for outcomes to occur; and H: Adequacy of follow up of cohorts.
Patient characteristics
Older patients, had comparable preoperative comorbidity compared with younger patients, except for ASA score and clinical tumor stage (older patients had more clinical stage II cancer, while younger patients had more clinical stage III cancer) and neoadjuvant therapy (less often in older patients). The extend of resection (transthoracic or transhiatal) was comparable between both groups, but more older patients underwent minimally invasive esophagectomy (Table (Table22).
Table 2
Baseline characteristics of included studies (age threshold 75 Years)
| Variable | Young | Old | Studies | P value | |||
| No. (% of total) | 4195 | 84.1% | 796 | 15.9% | 8 | ||
| n | % | n | % | n | % | ||
| Gender | < 0.001 | ||||||
| Male | 3572 | 85.1 | 623 | 78.3 | |||
| Female | 520 | 12.4 | 132 | 16.6 | |||
| Unknown | 103 | 2.5 | 41 | 5.2 | 1 | 12.5 | |
| Charlson comorbidity index | 0.058 | ||||||
| 0 | 202 | 4.8 | 38 | 4.8 | |||
| 1 | 90 | 2.1 | 28 | 3.5 | |||
| 2 | 65 | 1.5 | 23 | 2.9 | |||
| 3 | 0 | 0.0 | 0 | 0.0 | |||
| 4 | 0 | 0.0 | 0 | 0.0 | |||
| 5 | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 3838 | 91.5 | 707 | 88.8 | 7 | 87.5 | |
| Respiratory comorbidity | 0.272 | ||||||
| Yes | 43 | 1.0 | 8 | 1.0 | |||
| No | 361 | 8.6 | 104 | 13.1 | |||
| Unknown | 3791 | 90.4 | 684 | 85.9 | 6 | 75.0 | |
| Cardiac/Cardiovascular comorbidity | 0.244 | ||||||
| Yes | 88 | 2.1 | 28 | 3.5 | |||
| No | 316 | 7.5 | 84 | 10.6 | |||
| Unknown | 3791 | 90.4 | 684 | 85.9 | 6 | 75.0 | |
| Diabetes mellitus | 0.005 | ||||||
| Yes | 17 | 0.4 | 10 | 1.3 | |||
| No | 316 | 7.5 | 60 | 7.5 | |||
| Unknown | 3862 | 92.1 | 726 | 91.2 | 6 | 75.0 | |
| Renal insufficiency | 0.258 | ||||||
| Yes | 1 | 0.0 | 2 | 0.3 | |||
| No | 99 | 2.4 | 48 | 6.0 | |||
| Unknown | 4095 | 97.6 | 746 | 93.7 | 7 | 87.5 | |
| Liver cirrhosis | - | ||||||
| Yes | 0 | 0.0 | 0 | 0.0 | |||
| No | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 4195 | 100.0 | 796 | 100.0 | 8 | 100.0 | |
| ASA | < 0.001 | ||||||
| I | 164 | 3.9 | 13 | 1.6 | |||
| II | 598 | 14.3 | 117 | 14.7 | |||
| III | 194 | 4.6 | 66 | 8.3 | |||
| IV | 6 | 0.1 | 0 | 0.0 | |||
| V | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 3233 | 77.1 | 600 | 75.4 | 5 | 62.5 | |
| Tumor location in the esophagus | 0.239 | ||||||
| Cervical esophagus | 0 | 0.0 | 0 | 0.0 | |||
| Upper third | 25 | 0.7 | 4 | 0.5 | |||
| Middle third | 253 | 6.6 | 71 | 8.9 | |||
| Lower third | 79 | 2.1 | 14 | 1.8 | |||
| Junction | 0 | 0.0 | 0 | 0.0 | |||
| Cardia | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 0 | 0.0 | 0 | 0.0 | |||
| Unknown1 | 3838 | 100.0 | 707 | 88.8 | 7 | 87.5 | |
| Histological type | 0.022 | ||||||
| Adenocarcinoma | 2956 | 70.5 | 597 | 75.0 | |||
| Squamous cell carcinoma | 442 | 10.5 | 89 | 11.2 | |||
| Other | 21 | 0.5 | 0 | 0.0 | |||
| Unknown1 | 1 | 0.0 | 2 | 0.3 | |||
| Unknown | 775 | 18.5 | 108 | 13.6 | 2 | 25.0 | |
| cTNM | 0.039 | ||||||
| 0 | 56 | 1.3 | 4 | 0.5 | |||
| I | 234 | 5.6 | 40 | 5.0 | |||
| II | 525 | 12.5 | 94 | 11.8 | |||
| III | 547 | 13.0 | 83 | 10.4 | |||
| IV | 131 | 3.1 | 14 | 1.8 | |||
| Unknown1 | 70 | 1.7 | 3 | 0.4 | |||
| Unknown | 2632 | 62.7 | 558 | 70.1 | 4 | 50.0 | |
| pTNM | 0.648 | ||||||
| 0 | 1 | 0.0 | 0 | 0.0 | |||
| I | 36 | 0.9 | 21 | 2.6 | |||
| II | 20 | 0.5 | 9 | 1.1 | |||
| III | 36 | 0.9 | 19 | 2.4 | |||
| IV | 7 | 0.2 | 1 | 0.1 | |||
| Unknown1 | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 4095 | 97.6 | 746 | 93.7 | 7 | 87.5 | |
| Complete pathological response | 0.793 | ||||||
| Yes | 60 | 1.4 | 16 | 2.0 | |||
| No | 297 | 7.1 | 73 | 9.2 | |||
| Unknown | 3838 | 91.5 | 707 | 88.8 | 7 | 87.5 | |
| Neoadjuvant therapy | < 0.001 | ||||||
| Yes | 714 | 17.0 | 120 | 15.1 | |||
| No | 527 | 12.6 | 146 | 18.3 | |||
| Unknown | 2954 | 70.4 | 530 | 66.6 | 3 | 37.5 | |
| Resection type | - | ||||||
| Transthoracic | 590 | 14.1 | 109 | 13.7 | |||
| Transhiatal | 0 | 0.0 | 0 | 0.0 | |||
| Unknown | 3605 | 85.9 | 687 | 86.3 | 6 | 75.0 | |
| Surgical technique | 0.046 | ||||||
| Ivor lewis | 472 | 11.3 | 98 | 12.3 | |||
| McKeown | 17 | 0.4 | 1 | 0.1 | |||
| Orringer | 0 | 0.0 | 0 | 0.0 | |||
| Other | 101 | 2.4 | 10 | 1.3 | |||
| Unknown | 3605 | 85.9 | 687 | 86.3 | 6 | 75.0 | |
| Surgical approach | < 0.001 | ||||||
| Open | 705 | 16.8 | 116 | 14.6 | |||
| Minimally invasive | 525 | 12.5 | 129 | 16.2 | |||
| Hybrid | 11 | 0.3 | 21 | 2.6 | |||
| Unknown | 2954 | 70.4 | 530 | 66.6 | 3 | 37.5 |
Open in a separate window1Unknown: as stated in the included study. Only studies using an age threshold of 75 years to determine older patients are shown here. Regarding clinical and pathological tumor node metastasis stage, no particular tumor node metastasis edition was used.
Primary outcomes
The 90-d mortality rate was 9.05% in older patients and 3.92% in younger patients (95%CI = 1.10-5.56). In addition, the in-hospital mortality rate was 9.45% in older patients and 3.68% in younger patients (95%CI = 1.01-5.91). Anastomotic leak rates, rates of pulmonary and cardiovascular complications, and 30-d mortality rates were comparable between older and younger patients. Delirium was reported only once, therefore no meta-analysis was possible (Figures (Figures22 and and33).

Open in a separate windowFigure 2
Primary outcomes in forest plots. RR: Relative Risk; CI: Confidence Interval; age threshold = 75 years.

Open in a separate windowFigure 3
Funnel plots of primary outcomes. Age threshold = 75 years.
Secondary outcomes
Older and younger patients had comparable rates of chylothorax, pneumonia, hospital length of stay, 5-year overall survival, and 5-year disease-specific survival. Quality of life was not reported (Figure (Figure44).

Open in a separate windowFigure 4
Secondary outcomes in forest plots. RR: Relative risk; CI: Confidence interval. Age threshold = 75 years.
Subgroup analyses of studies on MIE
Two studies with 812 patients (151 older and 661 younger). From the primary outcomes, older patients showed comparable anastomotic leak, 30-d, 90-d, and in-hospital mortality rates. From the secondary outcomes, older patients showed comparable rates of chylothorax, and pneumonia (Table (Table33).
Table 3
Subanalysis of primary and secondary outcomes: open esophagectomy and minimally invasive esophagectomy
| Studies | Participants | Younger | Older | RR | 95%CI | P value | ||
| n | n | % | % | |||||
| Primary outcomes | ||||||||
| Anastomotic leak | ||||||||
| OE | 2 | 545 | 12.08 | 13.85 | 1.1307 | 0.2235 | 5.7199 | 0.5120 |
| MIE | 2 | 812 | 18.76 | 18.54 | 0.9996 | 0.1877 | 5.3243 | 0.9979 |
| 30-d mortality rate | ||||||||
| OE | 1 | 253 | 0.43 | 5.00 | 11.6500 | 0.7568 | 179.3310 | 0.0784 |
| MIE | 2 | 812 | 2.42 | 3.97 | 1.7320 | 0.0002 | 12493.0189 | 0.5761 |
| 90-d mortality rate | ||||||||
| OE | 2 | 403 | 0.60 | 2.86 | 6.8396 | 0.0279 | 1675.2496 | 0.1410 |
| MIE | 2 | 812 | 5.60 | 25.71 | 2.1859 | 0.2111 | 22.6353 | 0.1471 |
| In-hospital morality rate | ||||||||
| OE | 1 | 292 | 8.10 | 24.44 | 3.0189 | 1.5547 | 5.8620 | 0.0011 |
| MIE | 1 | 446 | 3.08 | 4.49 | 1.4586 | 0.4757 | 4.4730 | 0.5091 |
| Secondary outcomes | ||||||||
| Chylothorax | ||||||||
| OE | 1 | 292 | 3.24 | 2.22 | 0.6861 | 0.0879 | 5.3534 | 0.7193 |
| MIE | 1 | 446 | 8.12 | 7.87 | 0.9682 | 0.4385 | 2.1377 | 0.9363 |
| Pneumonia | ||||||||
| OE | 2 | 545 | 21.04 | 47.69 | 2.1531 | 0.0639 | 72.5068 | 0.2205 |
| MIE | 1 | 446 | 32.77 | 31.46 | 0.9600 | 0.6828 | 1.3497 | 0.8141 |
Standardized mean difference instead of relative risk. RR: Relative risk; 95%CI: 95% confidence interval. OE: Open esophagectomy; MIE: Minimally invasive esophagectomy.
Subgroup analyses of studies on OE
Two studies with 545 patients (65 older and 480 younger). From the primary outcomes, older patients had higher rates of in-hospital mortality, while anastomotic leak, 30-d and 90-d mortality rates were comparable. From the secondary outcomes, chylothorax and pneumonia rates were comparable between older and younger patients (Table (Table3).3).Go to:
DISCUSSION
We found that patients older than 75 years who underwent esophagectomy have a higher risk of 90-d and in-hospital mortality. Postoperative complications, 30-d mortality rate and survival were comparable between older and younger patients. In the subgroup of studies with MIE, older patients had comparable 30-d, 90-d and in-hospital mortality rates compared to younger patients.
Strengths of this review are the subgroup analyses of MIE and OE, the large number of patients that were included, and the fact that the studies included in this study were not used in previous meta-analyses. Another strength is the inclusion and separate analysis of different age thresholds to ensure that no studies were left out. These analyses did not show substantially different results with regard to the main analysis. The most important limitations of this review are the fact that only 1 prospective study was included, and only 2 studies which included patients that underwent MIE.
The higher 90-d mortality rates in patients aged 75 years and older found in this review correspond to results of earlier systematic reviews that used other age thresholds (70 and 80 years)[8-10]. More recently published studies confirm these results[27-29]. Literature shows, that older patients have a worse comorbidity status preoperatively compared with their younger counterparts, and as such have a higher risk of (severe) complications and mortality[30-33]. This is especially true, when considering that older patients with a similar comorbidity status as younger patients, have comparable short-term outcomes[13,34]. Our study, however, showed that the comorbidity status between patients aged < 75 and ≥ 75 was comparable, and this may be the result of lack of data, since it was not possible to obtain comorbidity rates for meta-analysis from most studies.
Subgroup analyses of OE studies showed increased in-hospital mortality rates in older patients, while studies including patients that underwent MIE showed comparable in-hospital, 30-d and 90-d mortality rates between older and younger patients. MIE might give surgeons opportunities to expand indications for curative surgery to older patients, because of the lower physical burden and risk of operation trauma of minimally invasive surgery on patients. Alternatively, it can be argued that studies with MIE used different selection criteria, as the two MIE studies in this review suggest[13,22].
The results from this review underline the importance of age as a predictor of mortality in patients undergoing esophagectomy for esophageal cancer, and this should be taken into account by clinicians. The results of this review also suggest that MIE might give older patients a chance at curative resection without a higher risk of mortality as compared to younger patients. In our view, however, it remains important to take comorbidity, fitness, frailty and patients’ views into consideration in addition to patient age and surgical approach.Go to:
CONCLUSION
In conclusion, patients aged 75 years or older undergoing curative esophagectomy for esophageal cancer have a higher risk of mortality. Minimally invasive esophagectomy may be important for minimizing mortality in older patients.
原文链接:http://www.xxwk.net/archives/477