The rationale for ambulatory extracorporeal membrane oxygenation (ECMO) is simple:
- (1)Upright patients who are ambulatory and socially interactive provide the most effective vehicle for clinical recovery or subsequent bridge to transplant.
- (2)No lung disease or pulmonary injury benefits from paralysis, sedation, and intubation with nonphysiological positive pressure ventilation.
The objective data for these simple observations are well established and include the traditional morbidities of ventilator-associated pneumonias, barotrauma as a consequence of positive pressure ventilation, the requirements of sedation and paralytics to facilitate permissive hypercapnea as a strategy to limit barotrauma, and the profound deconditioning of both respiratory and skeletal muscle because of “ventilated, bed-bound” care.Although the use of ambulatory ECMO is an extension of traditional extracorporeal technologies, it is more “goal directed” and dynamic. The multiple cannulation strategies common to ambulatory ECMO are designed to facilitate an extubated and ambulatory patient. It is neither new nor novel and is analogous to the ventricular assist technologies common to patients with acute cardiogenic shock and congestive heart failure. Early deployment of technology to resuscitate the sick patient rather than reanimate the moribund is always the goal of extracorporeal support technologies. Nonetheless, the “peripheral” hybrid technologies of venovenoarterial ECMO and the application of “central” cannulation with “oxyRVADs” (right ventricular assist device with an in-line oxygenator) can salvage the sickest patients with medically refractory end-stage lung disease to ambulatory status.The following description of cannulation strategies is “goal directed” and “case specific.” Each is designed to support ambulatory patients with distinct clinical needs. Hypercapnea, hypoxia, or cardiopulmonary collapse as a consequence of cor pulmonale require different strategies of initial ECMO deployment. The described techniques are not exhaustive. Any cannulation strategy that delivers an adequate cardiac output with adequate gas exchange in an ambulatory patient is effective. Clinical need determines cannulation strategy. “Thought algorithms” regarding the application of ECMO—why are you doing this and what do you hope to accomplish—and the deployment of ECMO—how do we do it and when do we try—are useful in establishing conceptual ground rules for device deployment.
Conclusions
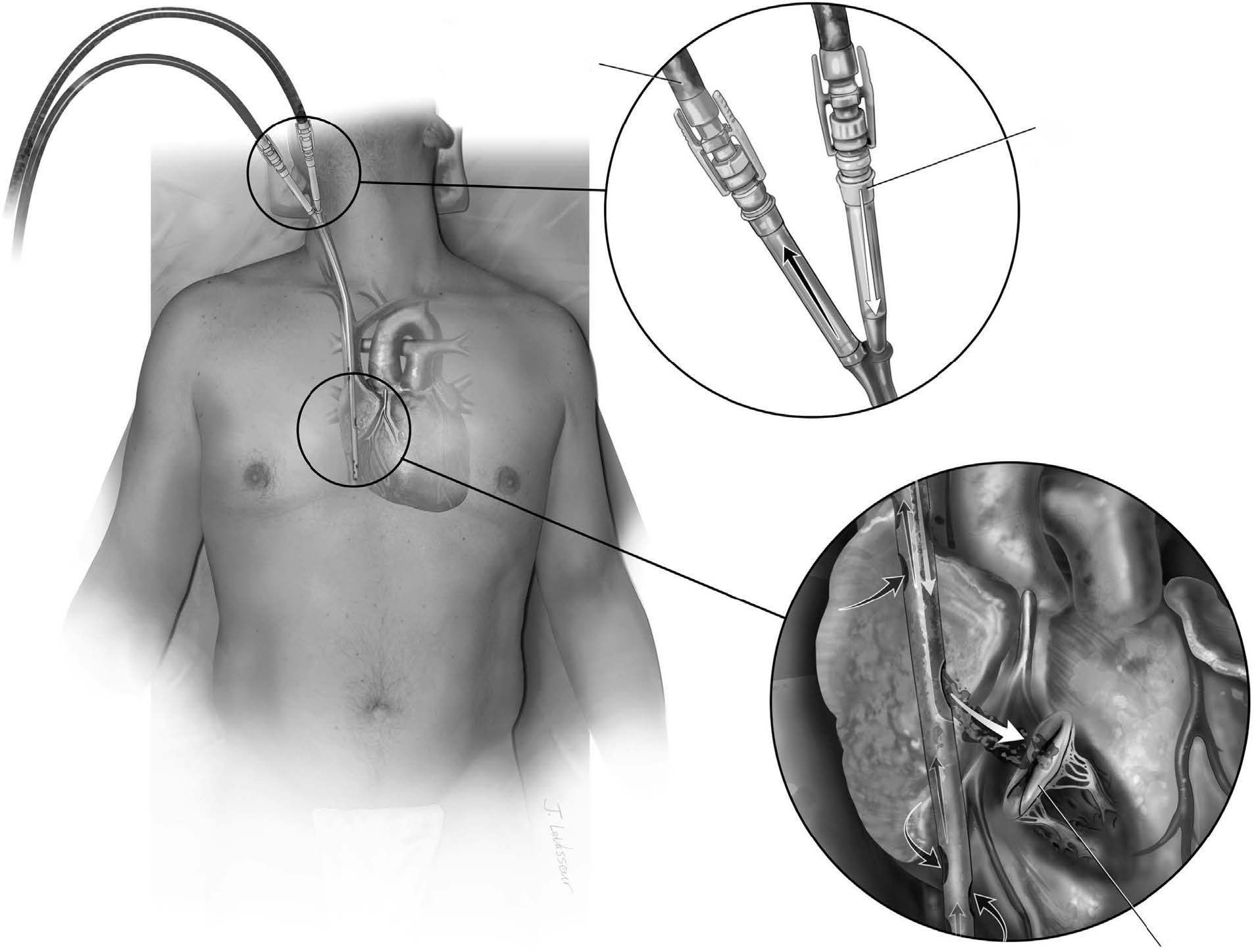
Although ECMO deployment is not technically difficult, many patients require multiple revisions of cannulation strategy to transition from mechanical ventilation to ambulation. A patient management algorithm is outlined here.1 In brief, appropriate patients are stabilized on femoral venoarterial ECMO (25 Fr venous and 17 Fr aortic). All patients undergo open tracheostomy at the time of ECMO deployment. All sedation and vasopressor support is weaned and patients with significant neurologic deficits are removed from ECMO support. The “awake” patient without echocardiography (ECHO) criteria for compromised ventricular function undergoes “PECLA triage” (pumpless extracorporeal lung assist) with removal of the centrifugal pump and antegrade flow from the femoral arterial cannula to the right atrium via a long venous cannula using native cardiac output. Patients with stable hemodynamics are switched to double-lumen venovenous ECMO (Fig. 1) for ambulation. Patients with any hemodynamic concerns are deployed on hybrid venovenoarterial ECMO using the axillary approach (Fig. 2). Patients with moderate to severe pulmonary hypertension have double-lumen venovenous ECMO deployed with an associated atrial septostomy to preload the underfilled left ventricle with membrane oxygenated blood ( Figs. 3 and 4). Patients with RV failure due to end-stage lung disease undergo central cannulation w ith an “oxyRVAD” configuration as an ambulatory bridge to transplant (Figure 5, Figure 6). Patients with mixed end-stage heart and lun g disease are placed on “walking bypass” (Fig. 7).
原文链接:http://www.xxwk.net/archives/1684